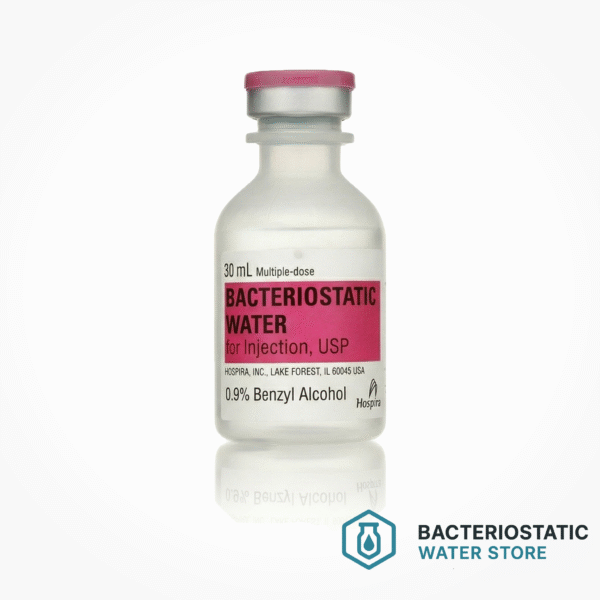
Hospira® Bacteriostatic Water for Injection, USP — 30 mL Multi-Dose Vial
Technical Data Sheet · Wholesale Procurement · Pfizer/Hospira Manufacturing
Composition, pH, and Preservation Standards
Bacteriostatic Water for Injection, USP is sterile water preserved with 0.9% (9 mg/mL) benzyl alcohol as a bacteriostatic agent. The inclusion of benzyl alcohol inhibits microbial growth between successive punctures, qualifying this product as a multi-dose diluent suitable for repeated access under aseptic conditions. Each vial is pyrogen-free and manufactured to United States Pharmacopeia monograph standards.
| Parameter | Specification | Standard / Method |
|---|---|---|
| Active ingredient | Water for Injection, USP | USP Monograph |
| Preservative | Benzyl alcohol 0.9% (9 mg/mL) | USP <51> Antimicrobial Effectiveness |
| pH range | 4.5 – 7.0 | USP <791> pH determination |
| Osmolarity | ~9 mOsmol/L (calculated) | USP <785> |
| Endotoxin limit | < 0.5 EU/mL | USP <85> Bacterial Endotoxins Test |
| Sterility | Sterile; pyrogen-free | USP <71> Sterility Tests |
| Container closure | Plastic flip-top vial, latex-free stopper | USP <381> / <661> |
| Fill volume | 30 mL nominal | USP <1> Injections and Implanted Drug Products |
Visual Markers of Genuine Hospira Supply
Authentic Hospira 30 mL vials can be verified through several manufacturing-specific markers. The flip-top cap is produced via a blow-fill-seal process with distinct beveled edges. The lot number and expiry date are heat-stamped directly into the plastic vial body—not applied as an inkjet label—making them resistant to abrasion. The printed label carries the Pfizer/Hospira trade dress with NDC 0409-3977-03 in machine-readable GS1-128 barcode format.
The 28-Day Rule for Punctured Multi-Dose Vials
Unopened vials should be stored at controlled room temperature, 20°C to 25°C (68°F to 77°F). Protect from light. USP <797> and CDC multi-dose vial policy require that once a vial is first punctured, it must be date-labeled and discarded within 28 days, regardless of the remaining volume. The bacteriostatic agent does not provide indefinite protection against contamination.
| Condition | Requirement | Regulatory Basis |
|---|---|---|
| Unopened storage | 20°C – 25°C; avoid freezing | USP <659> Packaging and Storage |
| Post-puncture use | 28 days maximum from first use | USP <797>; CDC MDV Policy |
| Post-puncture labeling | Date of first puncture recorded on vial | USP <797> §5.3 |
| Discard conditions | Turbidity, discoloration, visible particles | USP <1>; Hospira PI |
Aseptic Technique (USP <797> Guidelines)
- 1Verify vial integrity and expiry. Confirm NDC 0409-3977-03 and lot number against CoA documentation.
- 2Perform hand hygiene and don appropriate PPE (gloves, gown, mask) in accordance with institutional SOP.
- 3Disinfect the rubber stopper of the Hospira vial and the lyophilized vial with a sterile 70% isopropyl alcohol swab; allow 30 seconds to dry.
- 4Using a sterile luer-lock syringe, withdraw the required quantity. Direct the diluent stream against the interior wall of the vial to reduce foaming.
- 5Gently swirl — do not vortex — until reconstitution is complete. Label the vial with preparation date and record the date of first puncture on the Hospira vial.
| Contraindication | Detail |
|---|---|
| Neonates | Absolutely contraindicated due to fatal toxicity. |
| Hypersensitivity | Contraindicated for known benzyl alcohol hypersensitivity. |
| Direct IV Infusion | Not formulated for direct IV infusion; diluent use only. |
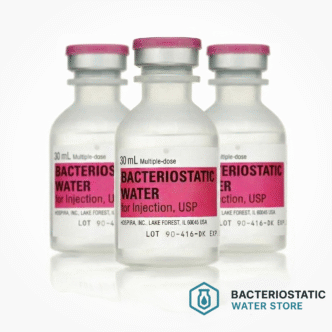
Bulk Ordering and Case Configurations
Institutional procurement is available in case quantitiesfor licensed healthcare facilities, pharmacy departments, and research institutions. Purchase orders should reference NDC 0409-3977-03.
| Pack Config | Units |
|---|---|
| Single Vial (30 mL) | 1 Unit |
| 3-Pack Value Bundle | 3 Units |
| 5-Pack Research Case | 5 Units |